| Condition | Very High Risk[A] Advise MUST NOT Fast | High Risk[A] Advise SHOULD NOT Fast | Low/Moderate Risk Decision to Not Fast Based on Discretion of Medical Opinion and Ability of the Individual to Tolerate Fast |
| Respiratory disease | - Those experiencing an acute exacerbation of their chronic lung disease
- Asthma/COPD sufferers at high risk of exacerbation whose preventative inhaler timings cannot be altered to a fasting compatible regimen
| - Poorly controlled lung disease with frequent exacerbations/hospital admissions
- Poorly controlled symptoms requiring frequent rescue inhaler and/or nebuliser use throughout the day
- Those receiving immunosuppressants for active lung disease
- Those receiving anti-fibrotic therapy
| - Well controlled asthma/COPD requiring intermittent inhaler use only
- Stable disease with infrequent exacerbations
- Those receiving immunosuppressants for stable disease (in remission)
|
| Cardiovascular disease[B] | - Advanced heart failure (optimal medical therapy, LVEF <35%, with class III–IV NYHA symptoms, ≥1 hospitalisation in the last 6 months due to decompensated heart failure and severely impaired functional capacity (e.g. 6-minute walk distance <300m)
- Severe pulmonary hypertension (defined as WHO/NYHA III–IV classification, right ventricular dysfunction and objective markers on right heart catheterisation (e.g. SvO2 <60%)
| - Recent acute coronary syndrome/myocardial infarction (<6 weeks)
- HOCM with significant left ventricular outflow tract gradient (e.g. peak gradient ≥50 mmHg)
- Severe valvular disease (defined by echocardiographic criteria)
- Severe heart failure without advanced features
- Poorly controlled arrhythmias (as defined by your specialist)
| - Hypertension
- Stable angina (episodes of angina are not occurring at rest or increasing significantly in frequency or severity)
- Mild HFrEF (LVEF ≥45%), moderate HFrEF (LVEF 35–45%), or HFpEF (diagnosed by a combination of symptoms, LVEF ≥45–50%, Heart Failure Association score, natriuretic peptide levels +/-imaging—refer to specialist confirmation)
- Intracardiac devices (pacemaker, ICD, CRT-D)
- Mild/mild–moderate valvular disease (as defined by echocardiographic criteria)
- Supraventricular tachycardias/atrial fibrillation/non-sustained ventricular tachycardia
- Mild/moderate pulmonary hypertension (pulmonary artery systolic pressure >25 mmHg without severe echocardiographic or right heart catheterisation features)
|
| CKD | - CKD patients in stage 4–5 with eGFR <30 ml/min
- Patients on all forms of haemodialysis and peritoneal dialysis
- Pregnant CKD patients
- Patients with inflammatory conditions of the kidney requiring immunosuppression
- CKD stage 3–5 patients with history of pre-existing cardiovascular disease
- CKD patients on tolvaptan
| - CKD patients in stage 3 (eGFR 30–60 ml/min)
- CKD patients with known electrolyte abnormalities
- Patients at risk of dehydration due to fluid restriction requirements or need for diuretics
- CKD patients in stage 1–3 on ACEi/ARB
| - CKD patients in stages 1–2 with stable kidney function
- CKD patients prone to urinary tract infections or stone formation
|
| Gastrointestinal disease | - Patients with established cirrhosis, especially Child-Pugh B and C
- Patients who are <6 months post liver transplant
- Patients with symptomatic active IBD
- Patients with significant acute or chronic diarrhoea
- Patients with high output ileostomy
| - Liver transplant patients taking tacrolimus are at high risk of renal toxicity if they become dehydrated. They are also at risk of rejection if adherence to immunosuppression medication is not maintained due to fasting
- Patients on prednisolone at doses >20 mg per day
| - Patients with stable chronic liver disease without cirrhosis
- Patients with stable chronic IBD in remission, including those on immunosuppressants
- Patients with peptic ulcer disease, reflux oesophagitis, and IBS
|
| Neurological disease | - Any condition predisposing to respiratory complications, e.g. bulbar weakness, neuromuscular disorders[C]
- Myasthenia gravis on regular pyridostigmine more than 3 times per day
- MND
- Poorly controlled epilepsy, on multiple antiepileptic medications, history of status epilepticus
- Parkinson’s disease requiring regular levodopa
- Neurodegenerative disorders with cognitive impairment
| - Epilepsy requiring a medication regimen incompatible with fasting which cannot be modified safely in time for the next Ramadan
- Myasthenia gravis on pyridostigmine 3 times daily or less
- Parkinson’s disease with low requirement for levodopa in younger patients
| - History of cerebrovascular disease, dependent on level of disability
- History of MS, dependent on level of disability
- Well-controlled epilepsy with a medication regime compatible with the length of fast
- Myasthenia gravis not requiring pyridostigmine or purely ocular
- Migraine
|
| Diabetes | - Poorly controlled type 1 diabetes
- Acute hyperglycaemic diabetes complications within 3 months prior to Ramadan (DKA, HHS)
- Disabling hypoglycaemia: severe hypoglycaemia within 3 months prior to Ramadan, hypoglycaemia unawareness, recurrent hypoglycaemic episodes
- Advanced macrovascular diabetic complications
- Type 2 diabetes requiring insulin (MDI/biphasic) with no prior experience of safe fasting[C]
- Chronic dialysis and CKD (stage 4 and 5)
- Pregnancy in pre-existing diabetes or GDM treated with insulin or SUs
- Acute illness
- Old age with ill health
| - Well-controlled type 1 diabetes
- Type 2 diabetes with sustained poor control (consider: HbA1c >75 mmol/mol for over 12 months)
- Type 2 diabetes requiring insulin (MDI/biphasic) with prior experience of safe fasting
- Type 2 diabetes on SGLT2 antagonists[C] (consider alternatives/stopping)
- Stable macrovascular diabetes complications
- CKD stage 3
- Pregnant women with type 2 diabetes or GDM on diet or metformin
- Comorbidities with additional risk factors
- Treatment with drugs that can affect cognitive function
- People with diabetes performing intense physical labour
| - Well-controlled type 2 diabetes (on one or more of the following therapies):
- diet and lifestyle
- metformin
- gliptins
- GLP-1 agonists
- glitazones
- acarbose
- second generation sulfonylurea[C] (moderate risk: regular BM monitoring advised)
- basal insulin[C] (moderate risk: regular BM monitoring advised)
|
| Adrenal disease | - Any of the following:
- multimorbidity: major organ system involvement
- diabetes mellitus on insulin treatment
- pituitary (diabetes) insipidus
- adrenal crises in the last 12 months
- untreated mineralocorticoid deficiency
- untreated TSH deficiency
- pregnancy (>28 weeks)[C]
| - Any of the following:
- recent diagnosis of steroid dependence within the last 12 months[C]
- no prior experience of fasting, or steroid alterations, or adjustments in Ramadan[C]
- aldosterone deficiency (i.e. on fludrocortisone or mineralocorticoid replacement)[C]
- pregnancy (<28 weeks)
| - Must meet ALL criteria:
- stable and well-controlled steroid insufficiency
- previous experience of fasting and risk assessments
- no significant comorbidities
- understanding of adjustment and changes to steroid dosing during fasting, when to terminate fasts, and sick day rules
- access to prednisolone 5 mg once daily or healthcare professional who can support prescriptions
- access to emergency (IM) hydrocortisone and understanding of how to use this
|
| Benign haematological disorders | - Sickle cell disease, including HbSS, HbSC, HbS/beta thalassaemia, HbSO, HbSD, and those prone to sickle cell crisis
- Cold haemagglutinin disease with ongoing haemolysis
- Amyloidosis with renal impairment
- Antiphospholipid syndrome with history of blood clots
- Paroxysmal nocturnal haemoglobinuria with active haemolysis or history of recurrent thrombosis
- Thrombophilias with history of recurrent thrombosis, despite being on anticoagulation
| - Warm autoimmune haemolytic anaemia with active haemolysis
- Other haemolytic anaemias with active haemolysis
- Clotting disorders like the thrombophilias with history of thrombosis
- Aplastic anaemia on immunosuppression
- Thrombophilia with a history of thrombosis within the last 3 months, and on anticoagulation
| - Thalassaemia carriers and sickle cell carriers who are not prone to crises
- Aplastic anaemia not on active treatment
- White cell disorders with low count
- Inherited bleeding disorders
- Immune thrombocytopenias in remission
- Thrombophilia with history of thrombosis on anticoagulation
|
| Haematological malignancies | - Patients requiring inpatient treatment for cancer or complications of cancer, e.g. acute leukemias, high grade lymphomas, aggressive/refractory myeloma
- Patients requiring inpatient treatment undergoing autologous or allogeneic stem cell transplantation or its complications
- Patients requiring inpatient treatment for complications of cancer treatment, e.g. neutropenic sepsis, severe vomiting, diarrhoea, pain, and other symptoms
- Newly diagnosed myeloma patients who are at risk of kidney injury
| - Patients taking tacrolimus or ciclosporin where risk of kidney injury is increased by dehydration
- Patients newly commenced on induction chemotherapy for haematological malignancies such as myeloma, lymphoma, chronic leukaemias, or experiencing significant side effects
- Patients receiving oral chemotherapy or targeted therapy, that:
- requires twice daily dosing
- must be taken with food
- is causing significant side effects
- Patients receiving a course of radiotherapy
- Patients who have undergone autologous or allogeneic transplantation within the last 6 months
- Patients receiving treatment for post-transplant complications such as GVHD
| - Patients receiving oral chemotherapy or targeted therapy, if:
- on a once daily dosing regimen
- drug pharmacokinetics allow fasting
- well established (>3 cycles) on treatment
- not experiencing significant side effects
- Patients receiving outpatient parenteral chemotherapy beyond induction phase (except on drug administration days) if:
- well established on treatment
- no/few manageable side effects
- Patients on parenteral maintenance immunotherapies with no/few manageable side effects, e.g. rituximab, obinutuzumab
- Outpatients with haematological cancers who are not receiving any active treatment and are on active surveillance only, e.g. MGUS, chronic leukaemias, low grade lymphomas
- Patients with previously treated cancers who are currently in remission and on active surveillance
|
| Rheumatological disease | - Active SLE with renal involvement
- Active vasculitis with renal involvement
- Low eGFR secondary to connective tissue diseases/vasculitis
- Scleroderma leading to pulmonary hypertension
| - Uncontrolled gout
- Higher dose of steroids >20 mg/day[C]
| - Rheumatological conditions in remission e.g. rheumatoid arthritis, polymyalgia rheumatica, connective tissue diseases, and vasculitis
- Osteoarthritis
- Osteoporosis
- Sjogren’s syndrome
- Well controlled gout
|
| Obesity | - BMI >40kg/m2 with any of the following:
- established end-organ cardiovascular disease (e.g. previous myocardial injury, cardiac failure, previous CVA/TIA)
- advanced CKD (stage 4–5)
- advanced chronic pulmonary diseases
- severe obstructive sleep apnoea
| - BMI >40kg/m2 with complicated metabolic syndrome and related complications e.g. those associated with high risk conditions (diabetes, hypertension, dyslipidemia, PCOS, hypothyroidism)
| - BMI >40 kg/m2 with stable non-metabolic comorbidities (e.g. osteoarthritis, fibromyalgia)
- Simple obesity without any comorbidities
|
| Pregnancy[D] | - Pregnancy with severe underlying maternal health conditions
- Complicated pregnancy
| - Uncomplicated pregnancy in an otherwise healthy woman in first trimester
- Pregnancy with moderately severe underlying maternal health conditions
| - Uncomplicated pregnancy in an otherwise healthy woman beyond first trimester
- Pregnancy with mild/well-controlled underlying maternal health conditions
|
| Organ transplants | - SOT recipients who underwent a transplant in the last 6 months
- Patients on twice daily immunosuppression
- Pregnant transplant patients
- Transplant patients diagnosed with new onset diabetes post-transplant requiring twice daily oral hypoglycaemics or insulin treatment
- Kidney transplant recipients with reduced kidney function (eGFR <30 ml/min)
- Patients with unstable graft function, rejection episodes, and opportunistic infections
| - Kidney transplant recipients with reduced kidney function (eGFR 60–30 ml/min)
- Heart, lung, liver, small bowel, pancreas, and multi-organ transplant recipients with reduced graft function
- Patients at risk of dehydration due to fluid restriction requirements, need for diuretics, or if they would be unable to meet their daily fluid intake requirement set by their transplant team
| - Transplant patients not in the above categories. The authors advise patients to discuss the suitability of fasting and monitoring necessary with their relevant transplant teams
|
| Solid tumours | - Patients on clinical trials
- Patients requiring inpatient treatment for cancer (or complications of cancer)
- Patients undergoing radical radiotherapy (especially head and neck, CNS, and upper GI malignancies)
- Patients receiving immunotherapy
| - Patients receiving intravenous chemotherapy who:
- have newly commenced (cycles 1–2) their treatment regimen
- are experiencing significant side effects
- Patients receiving oral chemotherapy or targeted therapy:
- that requires twice daily dosing
- that must be taken with food
- who are experiencing significant side effects
- Patients receiving a course of radiotherapy (with or without chemotherapy)
- Patients immediately following cancer surgery
| - Patients receiving oral chemotherapy or targeted therapy, if:
- they are on a once daily dosing regimen
- the drug pharmacokinetics allow it to be taken while fasted
- they are well established on treatment
- they have no/few manageable side effects
- Patients receiving intravenous chemotherapy, if:
- they are well established (cycle 3 or beyond) on their treatment regimen
- they have no/few manageable side effects
- Patients on intravenous maintenance therapies (e.g. trastuzumab, bevacizumab) with no/few manageable side effects
- Patients on endocrine therapy or androgen deprivation therapies with no/few manageable side effects
- Patients receiving radiotherapy for skin cancer or breast cancer (if otherwise well)
- Patients receiving palliative (single fraction) radiotherapy (if otherwise well)
- Patients under cancer surveillance, who are more than 3 months beyond completion of cancer therapies (including surgery) and have recovered sufficiently
|
| Mental health | - Anorexia/bulimia nervosa with purging by vomiting; severe laxative abuse
- Severe substance dependence disorder where stopping regimen may cause harm
- Medication dosing interval shorter than fasting hours, and necessary to prevent relapse/harm
- Poorly controlled SMI disorders (including clozapine use)
- Risk of electrolyte imbalance (e.g. lithium or metformin) or medication out of range
| - Stable bipolar/psychosis with medication regime compatible with fasting hours, >6 months since relapse. Monitor during Ramadan
| - Mild mental health illness not affecting functioning
- Well-controlled mental illness (no relapses in previous 12 months) with previous history of safe fasting
|
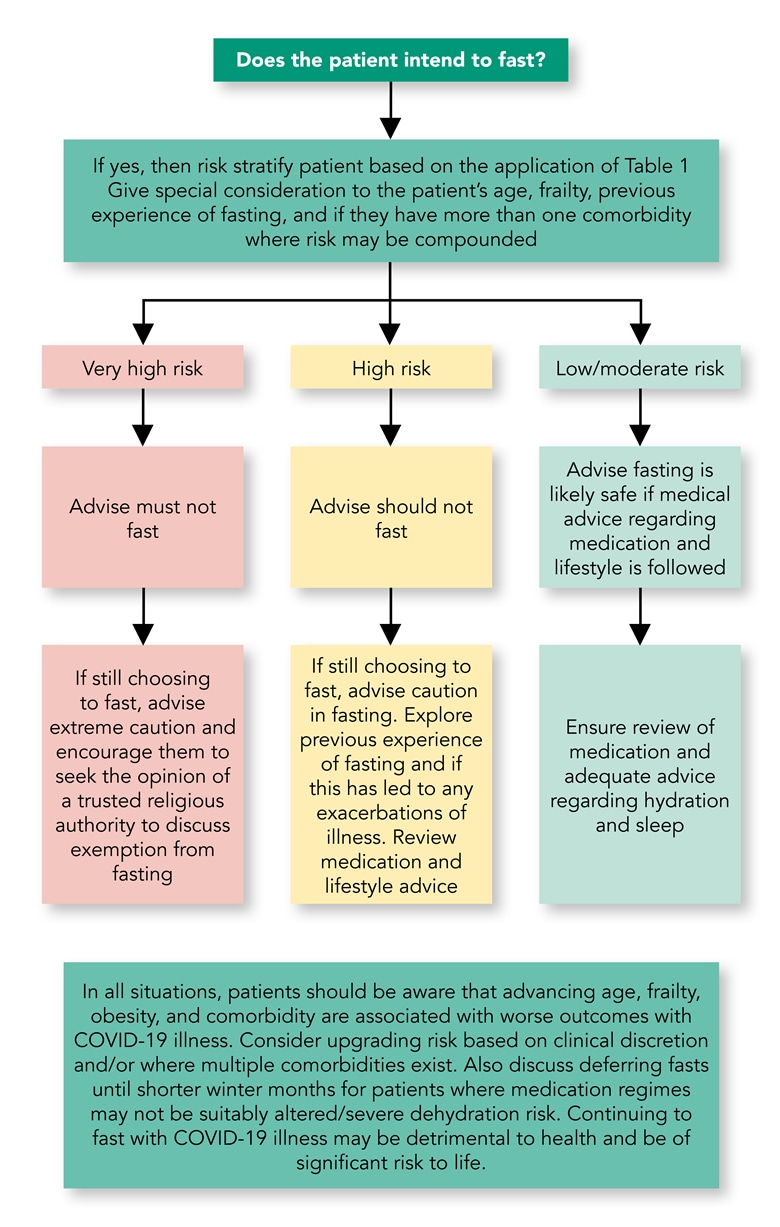
[A] If patients in these categories wish to fast, is shorter fasting in the winter a safe alternative? If this is not an option, or the patient is not willing to defer fasts and still wishes to fast, then they should be supported and should:- receive structured education (where appropriate)
- be followed by an appropriate specialist/primary care contact whilst fasting
- monitor their health regularly
- adjust medication dose, frequency, and timing as per recommendations
- be prepared to break the fast/abstain from fasting in case of adverse events
[B] Patients with GUCH and/or heart transplant must consult their specialist for an individual risk assessment
[C] Expert-recommended upgrading of risk due to COVID-19
[D] For breastfeeding, refer to the Muslim Council of Britain Ramadan Health Factsheet |
| ABN=Association of British Neurologists; ACEi=angiotensin-converting enzyme inhibitor; ARB=angiotensin receptor blocker; BM=capillary blood glucose; BMI=body mass index; CKD=chronic kidney disease; CNS=central nervous system; COPD=chronic obstructive pulmonary disease; CRT-D=cardiac resynchronisation therapy defibrillator; CVA=cerebral vascular accident; DKA=diabetic ketoacidosis; GDM=gestational diabetes mellitus; eGFR=estimated glomerular filtration rate; GI=gastrointestinal; GLP-1=glucagon-like peptide-1; GUCH=grown-up congenital heart disease; GVHD=graft-versus-host disease; HBA1c =haemoglobin A1c; HbS=sickle haemoglobin; HbSC=sickle cell haemoglobin SC; HbSD=sickle cell haemoglobin SD; HbSO=sickle cell haemoglobin SO; HbSS=sickle cell anaemia; HFpEF=heart failure with preserved ejection fraction; HFrEF=heart failure with reduced ejection fraction; HHS=hyperosmolar hyperglycaemic state; HOCM=hypertrophic obstructive cardiomyopathy; IBD=inflammatory bowel disease; IBS=irritable bowel syndrome; ICD=implantable cardioverter defibrillator; IM=intramuscular; LVEF=left ventricular ejection fraction; MDI=metered dose inhaler; MGUS=monoclonal gammopathy of undetermined significance; MND=motor neurone disease; MS=multiple sclerosis; NYHA=New York Heart Association; PCOS=polycystic ovary syndrome; SGLT2=sodium-glucose transport protein 2; SLE=systemic lupus erythematosus; SMI=serious mental illness; SOT=solid organ transplantation; SU=sulfonylurea; SvO2=venous oxygen saturation; TIA=transient ischaemic attack; TSH=thyroid stimulating hormone; WHO=World Health Organization |
- This is not an exhaustive list and is to be used for informative and shared decision making by healthcare professionals with patients. It does not form a directive. In all categories, patients should be advised to follow medical opinion due to probability of harm. Where appropriate, expert individualised medical advice must be sought before any decisions around fasting in Ramadan are made
- If a patient’s condition is not on this table and they have uncertainty or concerns about fasting, then they should seek medical advice before doing so. If this is not possible and they decide to fast, the advice given regarding terminating the fast should be followed
- The decision to fast is a personal decision for the individual concerned, who should be supported to achieve best possible outcomes
- Consider upgrading risk if unable to seek timely medical attention and make necessary changes to medication regimen, arrange baseline blood tests, or other preparation that usually precedes fasting
- Use the Rockwood clinical frailty score to assist with making assessments on risks of fasting in frail patients
- Ensure adequate hydration and nutrition
- Episodes of illness such as COVID-19 should be taken seriously and strong consideration should be given to breaking the fast, as the onset of illness can be rapid. Recovery from COVID-19 may also be prolonged. See Algorithm 1 and the section on acute illness for details
- Islamic jurists advise that any missed fasts should be made up in the future. However, if one’s health takes a permanent decline, such that even fasting during the winter period becomes unsafe or impossible, the fidyah would have to be paid. Patients should speak to a trusted religious authority before doing so.
|