Overview
This is a summary of the key recommendations from British Society of Gastroenterology guidelines for the management of iron deficiency anaemia in adults. This summary covers the diagnosis and management of iron deficiency anaemia (IDA) in adults in primary care settings in the UK. It refers to investigation and management in secondary care settings, but this summary of recommendations is primarily intended for primary healthcare professionals. For further information, refer to the full guideline.
Background and Definitions
Background
- IDA is common, and a major cause of morbidity worldwide
- IDA can be caused by a range of gastrointestinal (GI) pathologies including cancer, so GI investigation on an urgent basis should be considered with a new diagnosis of IDA without obvious explanation.
Definitions
- The British Society of Gastroenterology recommends that anaemia is defined as a haemoglobin (Hb) concentration below the lower limit of normal for the relevant population and laboratory performing the test
- Iron deficiency should be confirmed by iron studies prior to investigation. Serum ferritin is the single most useful marker of IDA, but other blood tests (for example, transferrin saturation) can be helpful if false-normal ferritin is suspected
- A good response to iron therapy (Hb rise 10 g/litre or greater within a 2-week timeframe) in patients with anaemia is highly suggestive of absolute iron deficiency, even if the results of iron studies are equivocal.
Initial Clinical Assessment
- Taking a detailed history is recommended, as it may provide important clues as to the cause(s) of IDA in the individual case
- Initial investigation of confirmed IDA should include urinalysis or urine microscopy, screening for coeliac disease (CD), and, in appropriate cases, endoscopic examination of the upper and lower GI tract
- CD is found in 3–5% of cases of IDA, and it is recommended that it should be routinely screened for serologically, or on small bowel biopsy at the time of gastroscopy
- Age, sex, Hb concentration, and mean cell volume are all independent predictors of risk of GI cancer in IDA and need to be considered as part of a holistic risk assessment. It follows that the cancer risk in iron deficiency without anaemia is low
- There are insufficient grounds at present to recommend faecal immunochemical testing for risk stratification in patients with IDA. The evidence base is evolving rapidly, however, and on that basis this guidance may change
- In men and postmenopausal women with newly diagnosed IDA, gastroscopy and colonoscopy should generally be the first-line GI investigations. In those not suitable for colonoscopy, computed tomography colonography is a reasonable alternative.
Table 1: Pathological Disorders Associated With IDA
| Chronic Blood Loss | |
| Digestive tract | Neoplastic—most commonly colonic adenocarcinomaInflammatory—e.g, peptic ulceration, IBD Vascular malformations—angiodysplasia Parasitic—e.g, hookworm |
| Genitourinary tract | Haematuria, pathological gynaecological bleeding—all cases, including malignancy |
| Respiratory tract | Recurrent epistaxis, haemoptysis—all causes |
| Malabsorption Syndromes | |
| Hypochlorhydria | Atrophic gastritis Helicobacter pylori infection Gastrectomy/gastric bypass Proton-pump inhibitors |
| Iron chelation | Tea, coffee, calcium, flavonoids, oxalates, phytates Wide range of antacids, Pica syndrome |
| Enteropathies | Coeliac disease Crohn’s disease NSAID enteropathy Rarer enteropathies, e.g, Whippie’s disease, bacterial overgrowth |
| Small bowel surgery | Small bowel resection/bypass |
| Genetic disorders | Iron-refractory IDA Divalent metal transporter 1 deficiency anaemia |
| Associated with the Anaemia of Chronic Disease | |
| Chronic heart failure | |
| Chronic kidney disease | |
| Chronic inflammatory disorders | E.g, rheumatoid arthritis, IBD |
| IBD=inflammatory bowel disease; IDA=iron deficiency anaemia; NSAID=non-steroidal anti-inflammatory drug | |
Follow-up, Recurrent IDA, and Further Evaluation
Follow-up and Recurrent IDA
- Hb levels normalise with iron replacement therapy (IRT) in most cases of IDA, but IDA recurs in a minority of those on long-term follow-up.
Further Evaluation of the Small Bowel
- In those with negative bidirectional endoscopy of acceptable quality and either an inadequate response to IRT or recurrent IDA, further investigation of the small bowel and renal tract is recommended to exclude other causes
- After negative capsule endoscopy of acceptable quality, further GI investigations need to be considered only if there is ongoing IDA after IRT
- Long-term IRT may be an appropriate strategy when the cause of recurrent IDA is unknown or irreversible.
Treatment of IDA
Algorithm 1: Overview of Treatment for IDA
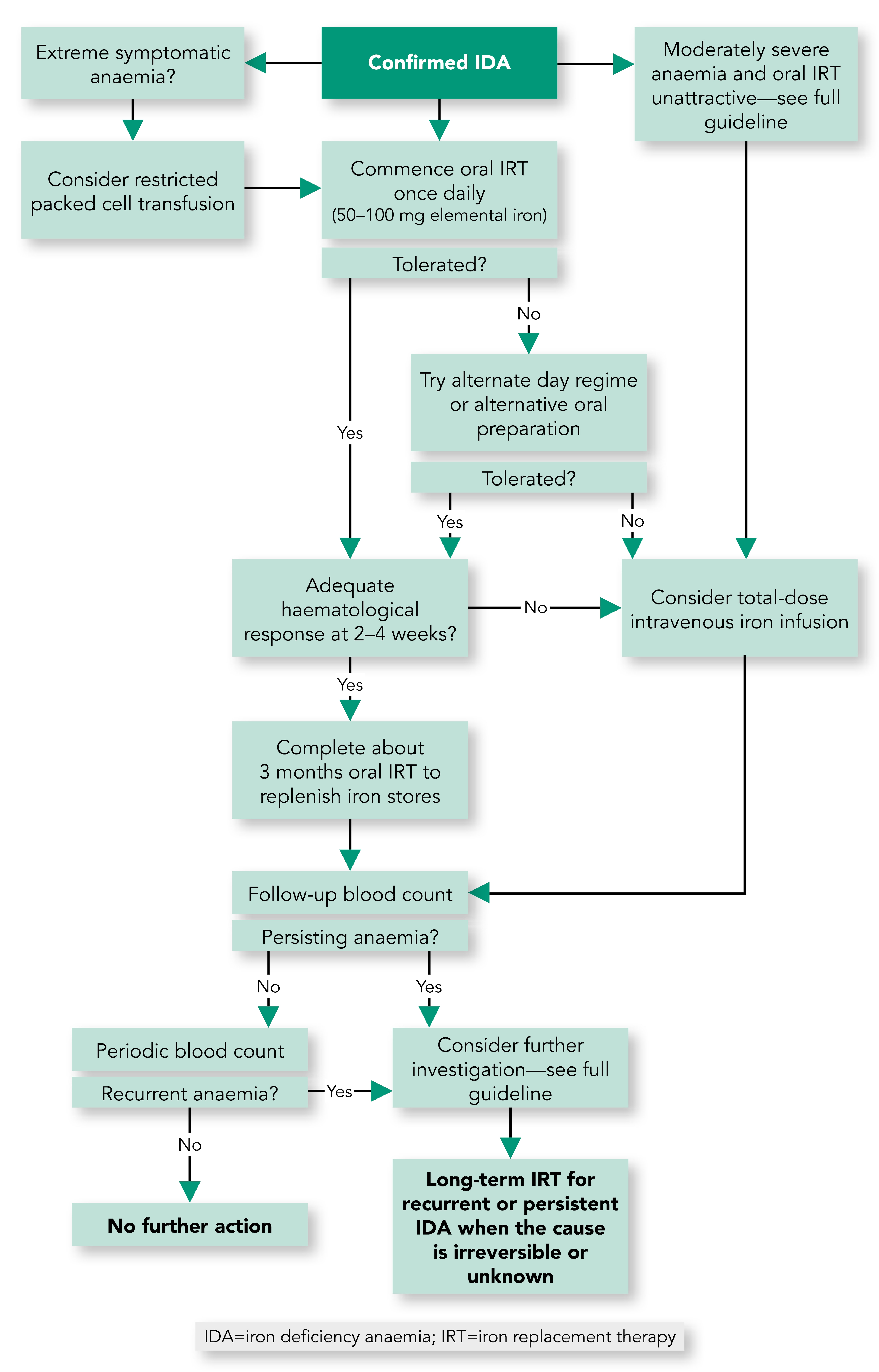
- IRT should not be deferred while awaiting investigations for IDA unless colonoscopy is imminent
- The initial treatment of IDA should be with one tablet per day of ferrous sulphate, fumarate, or gluconate. If not tolerated, a reduced dose of one tablet every other day, alternative oral preparations, or parenteral iron, should be considered
- Limited transfusion of packed red cells may on occasion be required to treat symptomatic IDA, in which case IRT is still necessary post-transfusion
- Patients should be monitored in the first 4 weeks for an Hb response to oral iron, and treatment should be continued for a period of around 3 months after normalisation of the Hb level, to ensure adequate repletion of the marrow iron stores
- Parenteral iron should be considered when oral iron is contraindicated, ineffective, or not tolerated. This consideration should be at an early stage if oral IRT is judged unlikely to be effective, and/or the correction of IDA is particularly urgent
- There is insufficient evidence to support invasive investigation in non-anaemic iron deficiency unless there are additional indications, but periodic blood count monitoring is suggested
- After the restoration of Hb and iron stores with IRT, it is recommended that the blood count should be monitored periodically (perhaps every 6 months initially) to detect recurrent IDA.
Special Situations
Young Women
- IDA is common in young women, and major contributory factors include menstrual losses, pregnancy, and poor dietary intake
- Underlying GI pathology is uncommon in young women with IDA, and so after screening for CD, further investigation is warranted only if there are additional clinical features of concern:
- age over 50, as age is a strong predictor of the risk of malignancy in IDA
- non-menstruating women—for example, following hysterectomy
- associated red flag symptoms, as outlined in NICE referral guidelines
- indications of a major genetic risk of GI pathology—for example, colorectal cancer affecting two first-degree relatives, or one first-degree relative affected before the age of 50 years
- recurrent or persistent IDA that appears disproportionate to other potential causes of iron deficiency, such as menstrual losses—accepting that this is usually a rather subjective judgement.
Young Men
- Confirmed IDA is uncommon in young men, but when found it warrants the same investigation as for elderly people (see later).
Elderly People
- Iron deficiency is common in elderly people, and is often multifactorial in aetiology
- It is recommended that the risks and benefits of invasive endoscopic and alternative investigation(s) are carefully considered in those with major comorbidities and/or limited performance status.
Specific Comorbidities
- Functional iron deficiency (FID) is a common contributory factor to the anaemia associated with advanced chronic kidney disease (CKD)
- Iron deficiency is common in chronic heart failure (CHF), and is often multifactorial
- Parenteral IRT may improve symptoms and quality of life in CHF with FID
- In the management of iron deficiency associated with CKD or CHF, reference to the appropriate specialist published guidelines is recommended
- IDA is a common manifestation of inflammatory bowel disease (IBD), particularly when the disease is active
- Intolerance and malabsorption of oral IRT can be particular problems in the treatment of IBD-associated IDA, and parenteral IRT may be required.
GI Surgery
- IDA is common following resection or bypass surgery involving the stomach and/or small bowel, including bariatric surgery
- In new presentation of IDA, a history of GI or bariatric surgery should not preclude a search for other causes of IDA.
Service Considerations
Algorithm 2: Algorithm for the Management of IDA
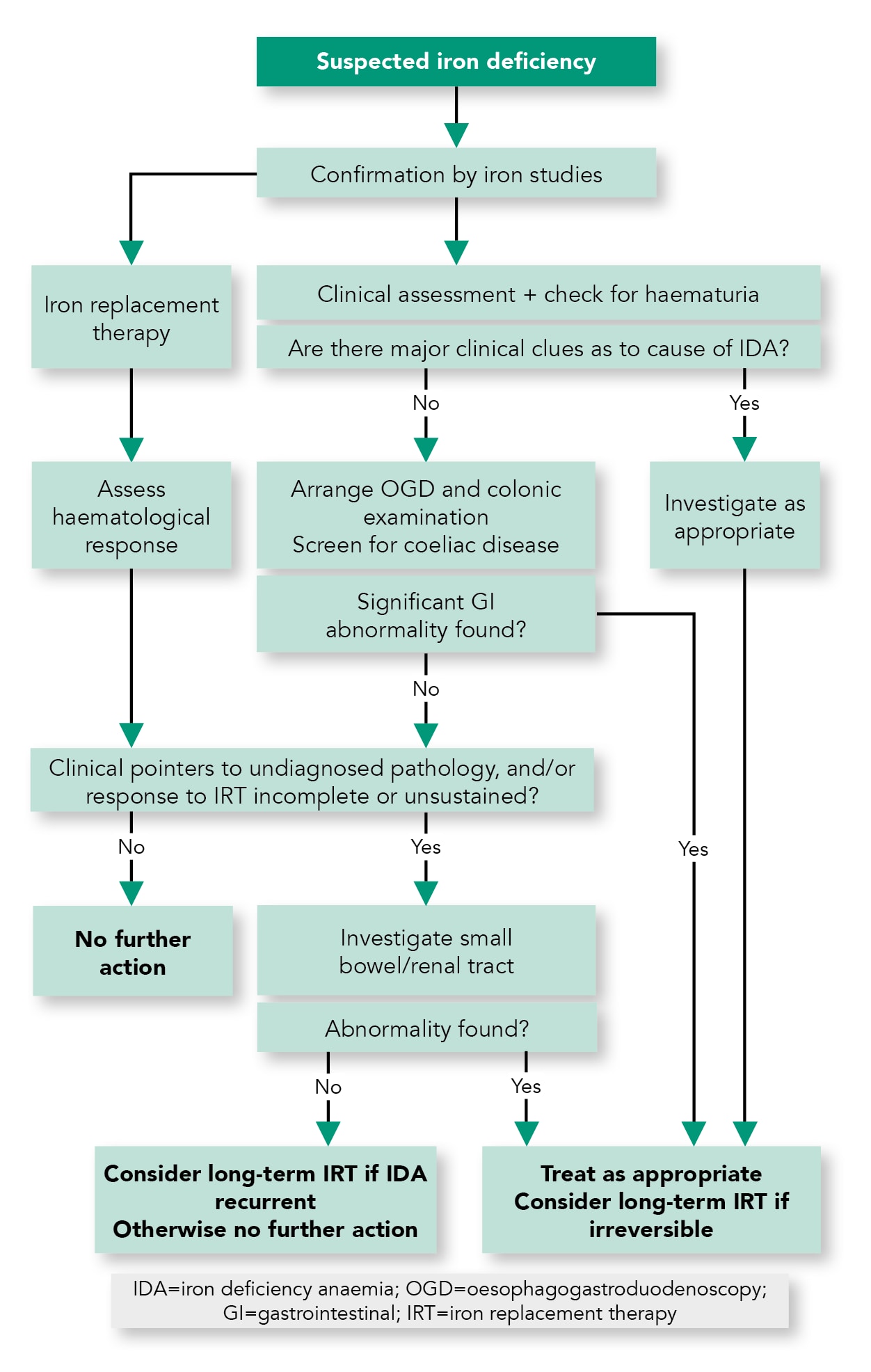
- All service providers should have clear points of referral and management pathways for patients with IDA (see Algorithm 2)
- To ensure efficient use of resources, IDA pathways should be delivered by a designated team led by a senior clinician
- Service providers should aim to have an ambulatory care base for the administration of parenteral iron.

