Overview
This pathway brings together multiple NICE clinical guidelines (CGs) and technology appraisals (TAs) into a single document, to support a consistent approach to the management of statin intolerance.
The guidelines and TAs included are:
- CG71: Familial hypercholesterolaemia: identification and management
- CG181: Cardiovascular disease: risk assessment and reduction, including lipid modification
- TA385: Ezetimibe for treating primary heterozygous-familial and non-familial hypercholesterolaemia
- TA393: Alirocumab for treating primary hypercholesterolaemia and mixed dyslipidaemia
- TA394: Evolocumab for treating primary hypercholesterolaemia and mixed dyslipidaemia
- TA694: Bempedoic acid with ezetimibe for treating primary hypercholesterolaemia or mixed dyslipidaemia
- TA733: Inclisiran for treating primary hypercholesterolaemia or mixed dyslipidaemia
- QS100: Cardiovascular risk assessment and lipid modification
- the Accelerated Access Collaborative lipid management algorithm.
Reflecting on your Learnings
Reflection is important for continuous learning and development, and a critical part of the revalidation process for UK healthcare professionals. Click here to access the Guidelines Reflection Record.
Statin Intolerance Pathway
Algorithm 1: Statin Intolerance Pathway
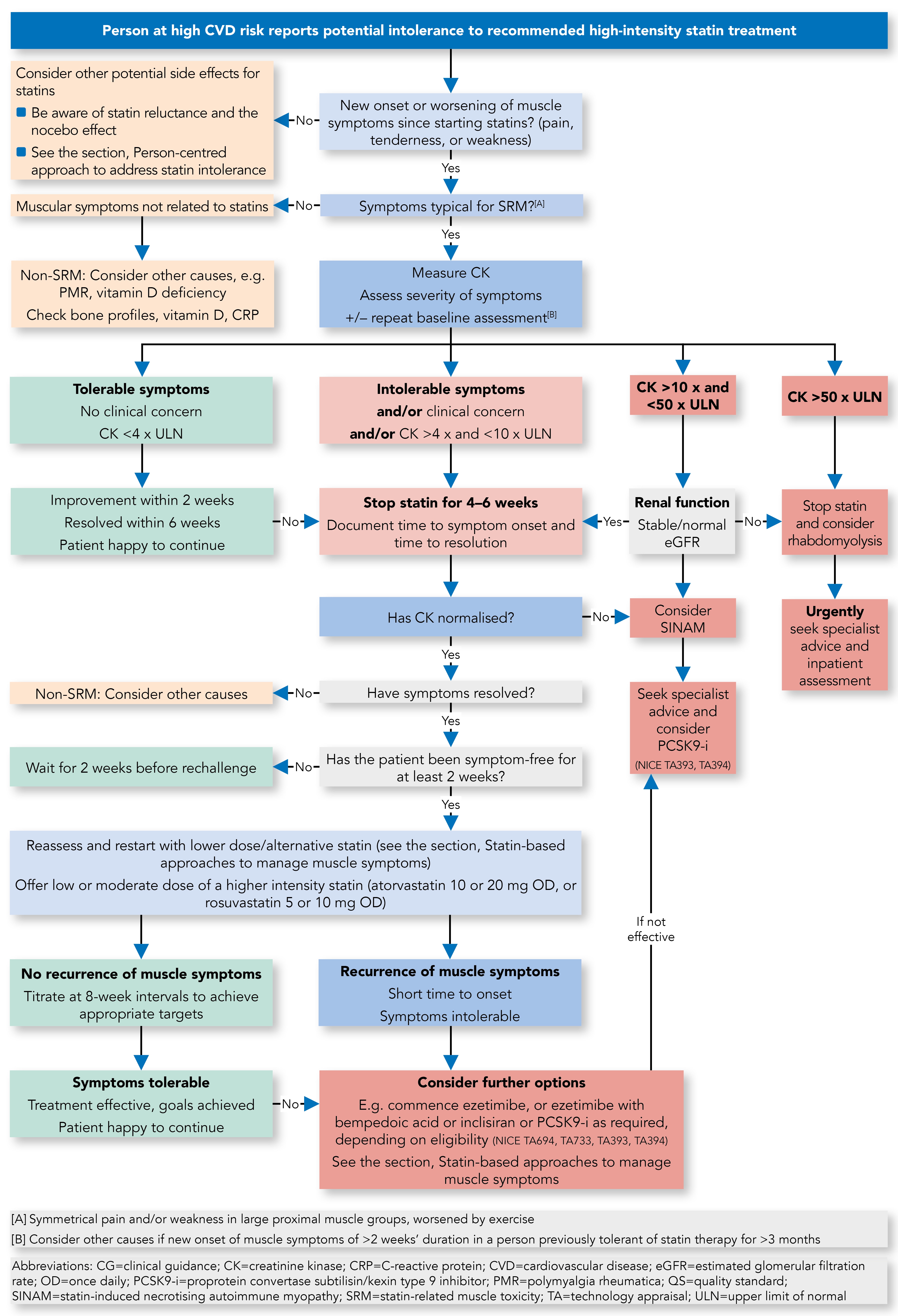
Definition of Statin Intolerance
- Intolerance to initial statin therapy is defined by NICE as the presence of clinically significant adverse effects that represent an unacceptable risk to the patient or that may reduce compliance with therapy
- Other definition: any adverse events (AEs) considered unacceptable by the patient, and/or some laboratory abnormalities, both attributed to statin treatment and leading to its discontinuation.
Statin-associated Muscle Symptoms
- Statin-associated muscle symptoms are one of the principal reasons for statin non-adherence and/or discontinuation. However, not all such symptoms should lead to a label of ‘statin intolerance’, as they may not be truly statin-related muscle toxicity (SRM) as demonstrated by resolution on dechallenge and recurrence with rechallenge.
Non-statin-related Musculoskeletal Symptoms
- If patients report symptoms that are not typical of SRM (for example, asymmetric distribution, failure to resolve with dechallenge despite normal creatine kinase [CK]), consider other musculoskeletal disorders (metabolic, degenerative, or inflammatory)—for example, vitamin D deficiency, polymyalgia rheumatica
- Check bone profile, vitamin D, C-reactive protein.
Considerations When Starting a Statin to Reduce Risk of SRM
- Check baseline thyroid, liver and renal function, any potential drug interactions, and avoid the highest doses in at-risk groups (See the section, Risk factors for SRM and statin intolerance)
- Ask the person if they have had persistent generalised unexplained muscle pain, whether associated or not with previous lipid-lowering therapy. If they have, measure CK. If CK levels are more than four times the upper limit of normal (ULN), do not start statin—investigation required
- Do not measure CK if person is asymptomatic
- Warn patients about AEs, specifically muscle symptoms. Advise people who are being treated with a statin to seek medical advice if they develop muscle symptoms (pain, tenderness, or weakness). If this occurs, measure CK.
Risk Factors for SRM and Statin Intolerance
Endogenous Factors
- Female gender
- Advanced age (over 75 years)
- Frailty (reduced lean body mass)
- History of muscle disorder or high CK
- Impaired renal or hepatic function
- Personal or family history of intolerance to lipid-lowering therapies
- Hypothyroidism.
Exogenous Factors
- Excessive alcohol intake
- High-intensity exercise
- Dehydration
- Drug interactions with statins (including herbal medicines)
- Vitamin D deficiency.
Classification of SRM
For a table outlining the classifications of SRM, refer to the full guideline.- SRM is a spectrum from myalgia to severe myopathy
- SRM 0: does not preclude statin therapy. Consider reducing starting dose
- SRM 1–3: manage according to pathway
- When SRM 4 is suspected, without evidence of impaired renal function, discontinue statin therapy immediately and refer for outpatient assessment. Assess and treat possible contributory factors and reassess the need for a statin. Intensify lifestyle modifications and consider alternative lipid lowering regimens
- If rhabdomyolysis (SRM 5) is suspected, immediately stop statins, and urgently refer to inpatient assessment and management, including intravenous rehydration as required to preserve renal function. Do not wait for measurement of urinary myoglobin. Post-recovery, manage as for SRM 4
- Statin-induced necrotising autoimmune myositis (SRM 6) should be suspected in patients with progressive muscle weakness and ongoing CK elevation despite statin withdrawal. It requires immunosuppressive treatment and avoidance of re-exposure to statins. Reassess the need for lipid lowering therapy—patients may be eligible for treatment with a PCSK9 inhibitor (NICE TA393 and TA394).
Person-centred Approach to Address Statin Intolerance
Initial Consultation
- Be aware of the ‘nocebo effect’[A] and ‘statin reluctance’[B]
- Reinforce healthy lifestyle habits (for example, exercise, reducing weight)
- Listen to the concerns of each patient
- Explain low-density lipoprotein cholesterol (LDL-C) targets and strategies to lower LDL-C/non-high-density lipoprotein (non-HDL-C)
- Discuss options to reduce LDL-C/non-HDL-C with pros and cons
- Explain the benefits of statins
- Evaluate and identify any risk factors and address them (for example, drug interactions)
- Work with patients to identify and agree best options and next steps.
Follow-up
- Follow up on agreed plan and address any issues/concerns
- Advise patients to contact you if they experience muscle symptoms
- Ongoing patient education and regular reviews help to address concerns around medicine safety and underline the importance of adherence.
Statin-based Approaches to Manage Muscle Symptoms
- Adopt a person-centred approach, as described in the previous section
- Therapy with a lower-dose statin is preferred to no statin
- Apply a repetitive ‘dechallenge’–‘rechallenge’ approach to establish if symptoms are caused by a statin(s) and the best statin regimen for each patient
- Switch to a different statin, or rechallenge with the same statin using a lower dose or frequency (intermittent dosages)
- For patients who do not tolerate statins on a daily basis, alternate-day or twice-weekly dosing is a good option
- Rosuvastatin and atorvastatin have longer half-lives, permitting their use on a non-daily regime
- Adding ezetimibe to a lower-dose statin may be better tolerated with robust reduction of LDL-C/non-HDL-C
- Once a new regime is tolerated, dose/frequency can be up-titrated slowly to achieve LDL-C/non-HDL-C goals with minimal or no muscle complaints
- It is important to note that cardiovascular benefits have not been proven for all of the above approaches, but any reduction of LDL-C/non-HDL-C is beneficial.
LDL-C-lowering Options for Patients With Genuine Statin Intolerance
- Refer to the Accelerated Access Collaborative lipid management algorithm
- Consider ezetimibe (NICE TA385) therapy as per algorithm
- Consider ezetimibe combined with bempedoic acid (NICE TA694), as per algorithm
- Consider inclisiran, if eligible, for treatment according to NICE TA733
- Consider PCSK9-i if eligible for treatment according to NICE TA393 and TA394.
Non-muscle-related Statin Side Effects
- May vary between different statins
- In clinical trials, some side effects often associated with statins are not statistically different from placebo
- Most commonly reported:
- gastrointestinal disturbance and asymptomatic increases in hepatic transaminases (alanine transaminase or aspartate aminotransferase)
- may affect up to one in 10 statin users
- Rarer side effects include:
- hepatotoxicity
- new onset type 2 diabetes (benefits outweigh risk; do not stop statin)
- renal insufficiency
- proteinuria
- neurocognitive and neurological impairments (no apparent link from randomised controlled trials)
- intracranial haemorrhage (conflicting evidence; benefits outweigh possible harm)
- interstitial lung disease
- pancreatitis
- skin disorders, including alopecia
- lupus-like reaction
- sleep disturbance
- headache
- dizziness
- fatigue
- depression
- sexual dysfunction
- Management:
- if symptoms appear statin-related, consider dechallenge and rechallenge or change to a different statin (for example, hydrophilic instead of lipophilic)
- liver enzyme abnormalities—minor increases in liver enzymes (less than twice ULN) may be seen within the first 3 months of statin therapy; temporary discontinuation and further assessment is warranted if levels exceed three times ULN.
Footnotes
[A] The nocebo effect is the negative expectation of the patient regarding a treatment, leading to reporting more negative effects even if they are prescribed a placebo.
[B] Statin reluctance is an attitudinal state of aversion to taking statins (often without prior exposure).

