The following Guidelines summary is on the assessment and management of heavy menstrual bleeding (menorrhagia). It aims to help healthcare professionals to investigate the cause of heavy periods that are affecting a woman’s quality of life and to offer the right treatments, taking into account the woman’s priorities and preferences.
This summary includes recommendations on investigations for the cause of, and the management of, heavy menstrual bleeding, as well as recommendations on history, physical examination, and tests, and useful information for women.
This guideline replaces NICE Clinical Guideline 44, and is the basis of NICE Quality Statement 47.
Recommendations on ulipristal acetate: In May 2021, NICE reinstated and amended recommendations on ulipristal acetate (Esmya) in line with updated Medicines and Healthcare products Regulatory Agency (MHRA) safety advice on the risk of serious liver injury with ulipristal acetate.
Impact of Heavy Menstrual Bleeding on Women
- Recognise that heavy menstrual bleeding (HMB) has a major impact on a woman’s quality of life, and ensure that any intervention aims to improve this rather than focusing on blood loss.
History, Physical Examination, and Laboratory Tests
History
- Take a history from the woman that covers:
- the nature of the bleeding
- related symptoms, such as persistent intermenstrual bleeding, pelvic pain, and/or pressure symptoms, that might suggest uterine cavity abnormality, histological abnormality, adenomyosis, or fibroids
- impact on her quality of life
- other factors that may affect treatment options (such as comorbidities or previous treatment for HMB)
- Take into account the range and natural variability in menstrual cycles and blood loss when diagnosing HMB, and discuss this variation with the woman. If the woman feels that she does not fall within the normal ranges, discuss care options
- If the woman has a history of HMB without other related symptoms, consider pharmacological treatment without carrying out a physical examination (unless the treatment chosen is levonorgestrel-releasing intrauterine system [LNG IUS][A]).
Physical Examination
- If the woman has a history of HMB with other related symptoms, offer a physical examination
- Carry out a physical examination before all investigations or LNG-IUS[A] fittings.
Laboratory Tests
- Carry out a full blood count test for all women with HMB, in parallel with any HMB treatment offered
- Testing for coagulation disorders (for example, von Willebrand’s disease) should be considered for women who:
- have had HMB since their periods started and
- have a personal or family history suggesting a coagulation disorder
- Do not routinely carry out a serum ferritin test for women with HMB
- Do not carry out female hormone testing for women with HMB
- Do not carry out thyroid hormone testing for women with HMB unless other signs and symptoms of thyroid disease are present.
Investigations for the Cause of Heavy Menstrual Bleeding
Algorithm 1: Heavy Menstrual Bleeding—Diagnosis
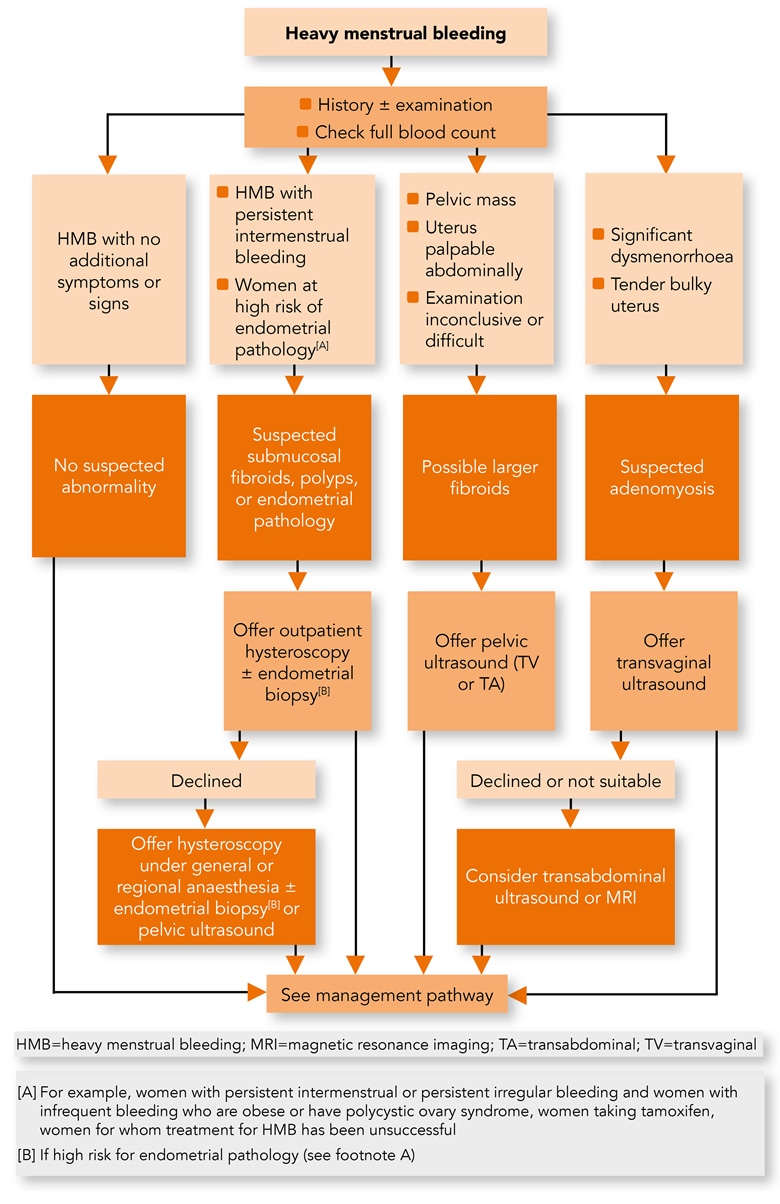
Before Starting Investigations
- Consider starting pharmacological treatment for HMB without investigating the cause if the woman’s history and/or examination suggests a low risk of fibroids, uterine cavity abnormality, histological abnormality, or adenomyosis
- If cancer is suspected, see NICE’s guideline on suspected cancer: recognition and referral.
Investigations
- Take into account the woman’s history and examination when deciding whether to offer hysteroscopy or ultrasound as the first-line investigation.
Women with Suspected Submucosal Fibroids, Polyps, or Endometrial Pathology
- Offer outpatient hysteroscopy to women with HMB if their history suggests submucosal fibroids, polyps, or endometrial pathology because:
- they have symptoms such as persistent intermenstrual bleeding or
- they have risk factors for endometrial pathology
- Ensure that outpatient hysteroscopy services are organised and the procedure is performed according to best practice, including:
- advising women to take oral analgesia before the procedure
- vaginoscopy as the standard diagnostic technique, using miniature hysteroscopes (3.5 mm or smaller)
- Ensure that hysteroscopy services are organised to enable progression to ‘see-and-treat’ hysteroscopy in a single setting if feasible
- Explain to women with HMB who are offered outpatient hysteroscopy what the procedure involves and discuss the possible alternatives
- If a woman declines outpatient hysteroscopy, offer hysteroscopy under general or regional anaesthesia
- For women who decline hysteroscopy, consider pelvic ultrasound, explaining the limitations of this technique for detecting uterine cavity causes of HMB
- Consider endometrial biopsy at the time of hysteroscopy for women who are at high risk of endometrial pathology, such as:
- women with persistent intermenstrual or persistent irregular bleeding, and women with infrequent heavy bleeding who are obese or have polycystic ovary syndrome
- women taking tamoxifen
- women for whom treatment for HMB has been unsuccessful
- Obtain an endometrial sample only in the context of diagnostic hysteroscopy. Do not offer ‘blind’ endometrial biopsy to women with HMB.
Women with Possible Larger Fibroids
- Offer pelvic ultrasound to women with HMB if any of the following apply:
- their uterus is palpable abdominally
- history or examination suggests a pelvic mass
- examination is inconclusive or difficult, for example in women who are obese.
Women with Suspected Adenomyosis
- Offer transvaginal ultrasound (in preference to transabdominal ultrasound or magnetic resonance imaging [MRI]) to women with HMB who have:
- significant dysmenorrhoea (period pain) or
- a bulky, tender uterus on examination that suggests adenomyosis
- If a woman declines transvaginal ultrasound or it is not suitable for her, consider transabdominal ultrasound or MRI, explaining the limitations of these techniques
- Be aware that pain associated with HMB may be caused by endometriosis rather than adenomyosis (see NICE’s guideline on endometriosis: diagnosis and management).
Other Diagnostic Tools
- Do not use saline infusion sonography as a first-line diagnostic tool for HMB
- Do not use MRI as a first-line diagnostic tool for HMB
- Do not use dilatation and curettage alone as a diagnostic tool for HMB.
Information for Women about Heavy Menstrual Bleeding and Treatments
- Provide women with information about HMB and its management. Follow the principles in the NICE guideline on patient experience in adult NHS services in relation to communication, information, and shared decision-making
- Provide information about all possible treatment options for HMB and discuss these with the woman (see the section, Management of Heavy Menstrual Bleeding). Discussions should cover:
- the benefits and risks of the various options
- suitable treatments if she is trying to conceive
- whether she wants to retain her fertility and/or her uterus.
Levonorgestrel-releasing Intrauterine System
- Explain to women who are offered an LNG-IUS:[A]
- about anticipated changes in bleeding pattern, particularly in the first few cycles and maybe lasting longer than 6 months
- that it is advisable to wait for at least six cycles to see the benefits of the treatment.
Impact of Treatments on Fertility
- Explain to women about the impact on fertility that any planned surgery or uterine artery embolisation may have, and if a potential treatment (hysterectomy or ablation) involves loss of fertility then opportunities for discussion should be made available
- Explain to women that uterine artery embolisation or myomectomy may potentially allow them to retain their fertility.
Endometrial Ablation
- Advise women to avoid subsequent pregnancy and use effective contraception, if needed, after endometrial ablation.
Hysterectomy
- Have a full discussion with all women who are considering hysterectomy about the implications of surgery before a decision is made. The discussion should include:
- sexual feelings
- impact on fertility
- bladder function
- need for further treatment
- treatment complications
- her expectations
- alternative surgery
- psychological impact
- Inform women about the increased risk of serious complications (such as intraoperative haemorrhage or damage to other abdominal organs) associated with hysterectomy when uterine fibroids are present
- Inform women about the risk of possible loss of ovarian function and its consequences, even if their ovaries are retained during hysterectomy.
Management of Heavy Menstrual Bleeding
Algorithm 2: Heavy Menstrual Bleeding—Management
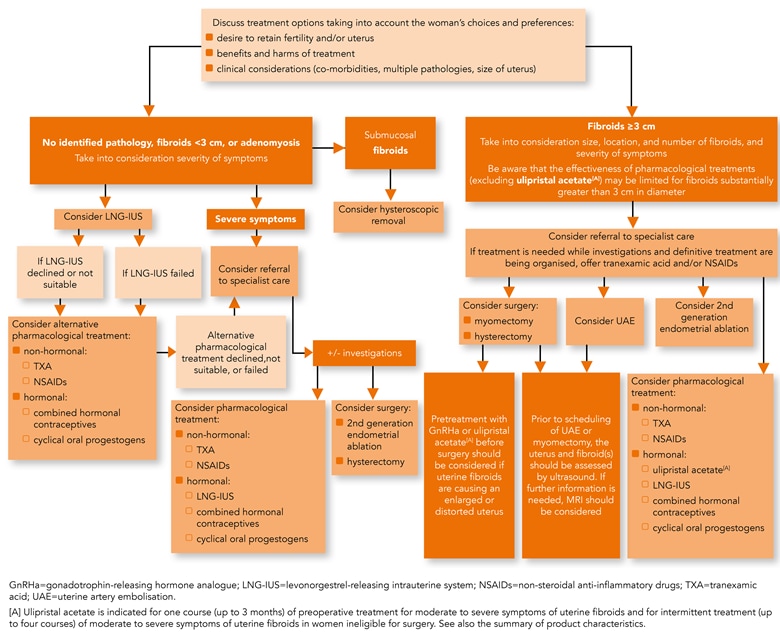
- When agreeing treatment options for HMB with women, take into account:
- the woman’s preferences
- any comorbidities
- the presence or absence of fibroids (including size, number, and location), polyps, endometrial pathology, or adenomyosis
- other symptoms such as pressure and pain.
Treatments for Women with no Identified Pathology, Fibroids less than 3 cm in Diameter, or Suspected or Diagnosed Adenomyosis
- Consider an LNG-IUS[A] as the first treatment for HMB in women with:
- no identified pathology or
- fibroids less than 3 cm in diameter, which are not causing distortion of the uterine cavity or
- suspected or diagnosed adenomyosis
- If a woman with HMB declines an LNG-IUS or it is not suitable, consider the following pharmacological treatments:
- non-hormonal:
- tranexamic acid
- non-steroidal anti-inflammatory drugs (NSAIDs)[B]
- hormonal:
- combined hormonal contraception[C]
- cyclical oral progestogens
- non-hormonal:
- Be aware that progestogen-only contraception may suppress menstruation, which could be beneficial to women with HMB
- If treatment is unsuccessful, the woman declines pharmacological treatment, or symptoms are severe, consider referral to specialist care for:
- investigations to diagnose the cause of HMB, if needed (see the section, Investigations for the Cause of HMB) taking into account any investigations the woman has already had and
- alternative treatment choices, including:
- pharmacological options not already tried
- surgical options:
- second-generation endometrial ablation
- hysterectomy
- For women with submucosal fibroids, consider hysteroscopic removal.
Treatments for Women with Fibroids of 3 cm or more in Diameter
- Consider referring women to specialist care to undertake additional investigations and discuss treatment options for fibroids of 3 cm or more in diameter
- If pharmacological treatment is needed while investigations and definitive treatment are being organised, offer tranexamic acid and/or NSAIDs[B]
- Advise women to continue using NSAIDs[B] and/or tranexamic acid for as long as they are found to be beneficial
- For women with fibroids of 3 cm or more in diameter, take into account the size, location, and number of fibroids, and the severity of the symptoms and consider the following treatments:
- pharmacological:
- non-hormonal:
- tranexamic acid
- NSAIDs[B]
- hormonal:
- LNG-IUS[A]
- combined hormonal contraception[C]
- cyclical oral progestogens
- ulipristal acetate (this is only indicated for some premenopausal women; see the following recommendations for more information)
- non-hormonal:
- uterine artery embolisation for fibroids
- surgical:
- myomectomy
- hysterectomy
- pharmacological:
- Only think about ulipristal acetate for the intermittent treatment of moderate to severe symptoms of uterine fibroids in premenopausal women if:
- surgery and uterine artery embolisation for fibroids are not suitable, for example, because the risks to a woman outweigh the possible benefits or
- surgery and uterine artery embolisation for fibroids have failed or
- the woman declines surgery and uterine artery embolisation for fibroids.
- Discuss with the woman the risks and possible benefits of intermittent treatment with ulipristal acetate
- advise that ulipristal acetate can be associated with serious liver injury leading to liver failure, and the signs and symptoms to look out for
- measure liver function before starting ulipristal acetate, monthly for the first two courses and once before each new treatment course when clinically indicated
- if there is no underlying liver injury, and surgery and uterine artery embolisation for fibroids are unsuitable or have failed, consider ulipristal acetate 5 mg (up to four courses) for premenopausal women with HMB and fibroids of 3 cm or more in diameter, particularly if the haemoglobin level is 102 g per litre or below
- if a woman shows signs and symptoms of liver failure, stop ulipristal acetate and perform liver function tests urgently
- Be aware that the effectiveness of pharmacological treatments for HMB may be limited in women with fibroids that are substantially greater than 3 cm in diameter.
Footnotes
[A] Note that this is an off-label use for some LNG-IUSs. See NICE’s information on prescribing medicines
[B] Note that this is an off-label use for NSAIDs. See NICE’s information on prescribing medicines
[C] Note that this is an off-label use for NSAIDs and some combined hormonal contraceptives. See the General Medical Council’s Prescribing guidance: prescribing unlicensed medicines for further information

