Overview
This Guidelines summary covers key recommendations for primary care on the assessment of chronic kidney disease (CKD). It aims to prevent or delay progression and reduce the risks of cardiovascular disease.
Key recommendations include: investigations for chronic kidney disease; classification of CKD in adults; frequency of monitoring; information and education for people with CKD; risk assessment, referral criteria and shared care; and pharmacotherapy.
View the related Guidelines summary, Chronic Kidney Disease and Anaemia, for recommendations on: diagnosing and assessing anaemia; managing anaemia; assessing and optimising erythropoiesis in people with anaemia; monitoring anaemia treatment; hyperphosphataemia in people with CKD stage 4 or 5; and other complications in adults.
For a complete set of recommendations, refer to the full guideline.
This guideline updates and replaces NICE Clinical Guideline 182 and NICE Guideline 8.
Visual Summary
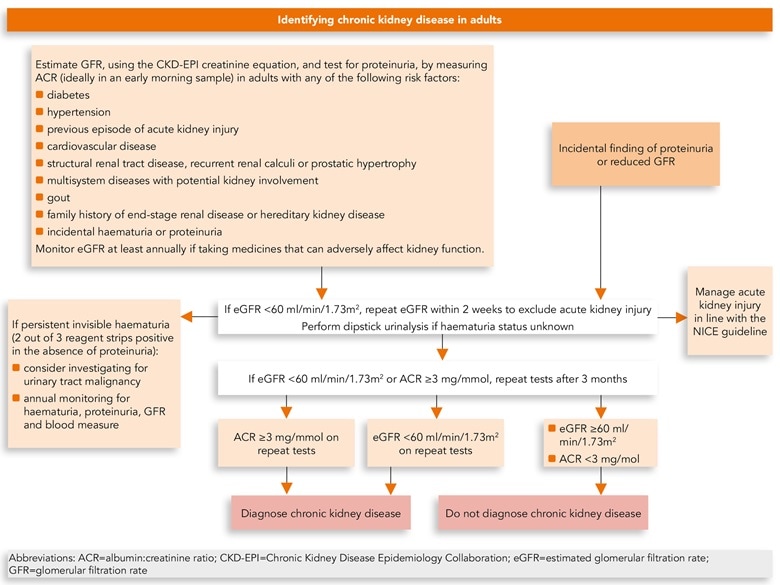
Algorithm 1: Identifying Chronic Kidney Disease in Adults
Investigations for Chronic Kidney Disease
Measuring Kidney Function
Creatinine-based Estimate of Glomerular Filtration Rate
- eGFRcreatinine may be less reliable in certain situations (for example, acute kidney injury, pregnancy, oedematous states, muscle wasting disorders, and in adults who are malnourished, who have higher muscle mass or use protein supplements, or who have had an amputation) and has not been well validated in certain ethnic groups (for example, black, Asian and other minority ethnic groups with CKD living in the UK).
- Interpret eGFRcreatinine with caution in adults with extremes of muscle mass, for example, in bodybuilders, people who have had an amputation or people with muscle wasting disorders. (Reduced muscle mass will lead to overestimation and increased muscle mass to underestimation of the GFR.)
- Advise adults not to eat any meat in the 12 hours before having a blood test for eGFRcreatinine. Avoid delaying the despatch of blood samples to ensure that they are received and processed by the laboratory within 12 hours of venepuncture.
Reporting and Interpreting GFR Values
- If eGFR is greater than 90 ml/min/1.73 m2, use an increase in serum creatinine concentration of more than 20% to infer significant reduction in kidney function.
- Interpret eGFR values of 60 ml/min/1.73 m2 or more with caution, bearing in mind that estimates of GFR become less accurate as the true GFR increases.
- Confirm an eGFR result of less than 60 ml/min/1.73 m2 in an adult not previously tested by repeating the test within 2 weeks. Allow for biological and analytical variability of serum creatinine (±5%) when interpreting changes in eGFR.
Investigations for Proteinuria
- Do not use reagent strips to identify proteinuria in children and young people.
- Do not use reagent strips to identify proteinuria in adults unless they are capable of specifically measuring albumin at low concentrations and expressing the result as an albumin:creatinine ratio (ACR).
- For the initial detection of proteinuria in adults, children and young people:
- use urine ACR rather than protein:creatinine ratio (PCR) because of the greater sensitivity for low levels of proteinuria
- check an ACR between 3 mg/mmol and 70 mg/mmol in a subsequent early morning sample to confirm the result.
- A repeat sample is not needed if the initial ACR is 70 mg/mmol or more.
- Regard a confirmed ACR of 3 mg/mmol or more as clinically important proteinuria.
- Measure proteinuria with urine ACR in the following groups:
- adults, children and young people with diabetes (type 1 or type 2)
- adults with an eGFR of less than 60 ml/min/1.73 m2
- adults with an eGFR of 60 ml/min/1.73 m2 or more if there is a strong suspicion of CKD
- children and young people without diabetes and with creatinine above the upper limit of the age-appropriate reference range.When ACR is 70 mg/mmol or more, PCR can be used as an alternative to ACR.
Incidental Finding of Proteinuria on Reagent Strips
- If unexplained proteinuria is an incidental finding on a reagent strip, offer testing for CKD using eGFRcreatinine and ACR.
Haematuria
- Use reagent strips to test for haematuria in adults, children and young people (see the last recommendation in the section, Investigations for proteinuria for people who should be tested for haematuria).
- Evaluate further for results of 1+ or higher.
- Do not use urine microscopy to confirm a positive result.
Managing Isolated Invisible Haematuria
- When there is the need to differentiate persistent invisible haematuria in the absence of proteinuria from transient haematuria, regard 2 out of 3 positive reagent strip tests as confirmation of persistent invisible haematuria.
- Persistent invisible haematuria, with or without proteinuria, should prompt investigation for urinary tract malignancy in appropriate age groups (see NICE’s guideline on suspected cancer: recognition and referral).
- Persistent invisible haematuria in the absence of proteinuria should be followed up annually with repeat testing for haematuria (see the previous two recommendations), proteinuria or albuminuria, GFR and blood pressure monitoring as long as the haematuria persists.
Who Should be Tested for CKD
- Monitor GFR at least annually in adults, children and young people who are taking medicines that can adversely affect kidney function, such as calcineurin inhibitors (for example, ciclosporin or tacrolimus), lithium or non-steroidal anti-inflammatory drugs (long-term chronic use of NSAIDs).
- Offer testing for CKD using eGFRcreatinine and ACR to adults with any of the following risk factors:
- diabetes
- hypertension
- previous episode of acute kidney injury
- cardiovascular disease (ischaemic heart disease, chronic heart failure, peripheral vascular disease or cerebral vascular disease)
- structural renal tract disease, recurrent renal calculi or prostatic hypertrophy
- multisystem diseases with potential kidney involvement, for example, systemic lupus erythematosus
- gout
- family history of end-stage renal disease (GFR category G5) or hereditary kidney disease
- incidental detection of haematuria or proteinuria.
- Offer testing for CKD using eGFRcreatinine and ACR to children and young people with any of the following risk factors:
- previous episode of acute kidney injury
- solitary functioning kidney.
- Consider testing for CKD using eGFRcreatinine and ACR in children and young people with any of the following risk factors:
- low birth weight (2,500 g or lower)
- diabetes
- hypertension
- cardiac disease
- structural renal tract disease or recurrent renal calculi
- multisystem diseases with potential kidney involvement, for example, systemic lupus erythematosus
- family history of end-stage renal disease (GFR category G5) or hereditary kidney disease
- incidental detection of haematuria or proteinuria.
- Do not use any of the following as risk factors indicating testing for CKD in adults, children and young people:
- age
- gender
- ethnicity
- obesity in the absence of metabolic syndrome, diabetes or hypertension.
- Monitor adults, children and young people for the development or progression of CKD for at least 3 years after acute kidney injury (longer for people with acute kidney injury stage 3) even if eGFR has returned to baseline.
- For guidance on ACR monitoring for children and young people with diabetes, see the NICE guideline on diabetes in children:
Classification of CKD in Adults
- CKD is classified according to estimated GFR (eGFR) and albumin:creatinine ratio (ACR) (see table 1 in the full guideline), using ‘G’ to denote the GFR category (G1 to G5, which have the same GFR thresholds as the CKD stages 1 to 5 recommended previously) and ‘A’ for the ACR category (A1 to A3), for example:
- A person with an eGFR of 25 ml/min/1.73 m2 and an ACR of 15 mg/mmol has CKD G4A2.
- A person with an eGFR of 50 ml/min/1.73 m2 and an ACR of 35 mg/mmol has CKD G3aA3.
- An eGFR of less than 15 ml/min/1.73 m2 (GFR category G5) is referred to as kidney failure.
- Classify CKD in adults using a combination of GFR and ACR categories. Be aware that:
- increased ACR is associated with increased risk of adverse outcomes
- decreased GFR is associated with increased risk of adverse outcomes
- increased ACR and decreased GFR in combination multiply the risk of adverse outcomes.
- Do not determine management of CKD solely by age.
Investigating the Cause of CKD and Determining the Risk of Adverse Outcomes
- Agree a plan to establish the cause of CKD during an informed discussion with the person with CKD, particularly if the cause may be treatable (for example, urinary tract obstruction, medicines that can adversely affect kidney function or glomerular disease).
- Use the person’s GFR and ACR categories (see table 1 in the full guideline) to indicate their risk of adverse outcomes (for example, CKD progression, acute kidney injury, all-cause mortality and cardiovascular events) and discuss this with them.
Indications for Renal Ultrasound in Adults
- Offer a renal ultrasound scan to all adults with CKD who:
- have accelerated progression of CKD (see the first recommendation in the section, Defining progression in adults)
- have visible or persistent invisible haematuria
- have symptoms of urinary tract obstruction
- have a family history of polycystic kidney disease and are older than 20
- have a GFR of less than 30 ml/min/1.73 m2 (GFR category G4 or G5)
- are considered by a nephrologist to need a renal biopsy.
- Advise adults with a family history of hereditary kidney disease about the implications of an abnormal result before a renal ultrasound scan is arranged for them.
Frequency of Monitoring
- If an adult, child or young person has CKD, or is at risk of it, agree the frequency of monitoring (eGFRcreatinine and ACR) with them (and their family members or carers, as appropriate), bearing in mind that CKD is not progressive in many people.
- When agreeing the frequency of monitoring, follow:
- the recommendations on patient views and preferences in NICE’s guideline on patient experience in adult NHS services
- NICE’s guideline on shared decision making.
- See the recommendations on when to refer adults and children and young people (see the section, Referral criteria) for specialist assessment.
- Use table 1 (below) to guide the minimum frequency of eGFRcreatinine monitoring, but tailor it according to:
- the underlying cause of CKD
- the rate of decline in eGFR or increase in ACR (but be aware that CKD progression is often non-linear)
- other risk factors, including heart failure, diabetes and hypertension
- changes to their treatment (such as renin–angiotensin–aldosterone system [RAAS] antagonists, NSAIDs and diuretics)
- intercurrent illness (for example acute kidney injury)
- whether they have chosen conservative management of CKD.
Table 1: Minimum Number of Monitoring Checks (eGFR:creatinine) Per Year for Adults, Children and Young People With or At Risk of Chronic Kidney Disease
Note: ACR monitoring should be individualised based on a person’s individual characteristics, risk of progression and whether a change in ACR is likely to lead to a change in management.| ACR Category A1: Normal to Mildly Increased (< 3 mg/mmol) | ACR Category A2: Moderately Increased (3–30 mg/mmol) | ACR Category A3: Severely Increased (> 30 mg/mmol) | |
|---|---|---|---|
| GFR Category G1: Normal and High (90 ml/min/1.73 m2 or over) | 0–1 | 1 | 1 or more |
| GFR Category G2: Mild Reduction Related to Normal Range for a Young Adult (60–89 ml/min/1.73 m2) | 0–1 | 1 | 1 or more |
| GFR Category G3a: Mild to Moderate Reduction (45–59 ml/min/1.73 m2) | 1 | 1 | 2 |
| GFR Category G3b: Moderate to Severe Reduction (30–44 ml/min/1.73 m2) | 1–2 | 2 | 2 or more |
| GFR Category G4: Severe Reduction (15–29 ml/min/1.73 m2) | 2 | 2 | 3 |
| GFR Category G5: Kidney Failure (under 15 ml/min/1.73 m2) | 4 | 4 or more | 4 or more |
| Abbreviations: ACR=albumin:creatinine ratio; GFR=glomerular filtration rate | |||
Defining Progression in Adults
- Define accelerated progression of CKD in adults as:
- a sustained decrease in GFR of 25% or more and a change in GFR category within 12 months or
- a sustained decrease in GFR of 15 ml/min/1.73 m2 per year.
- Take the following steps to identify the rate of progression of CKD:
- Obtain a minimum of 3 GFR estimations over a period of not less than 90 days.
- In adults with a new finding of reduced GFR, repeat the GFR within 2 weeks to exclude causes of acute deterioration of GFR. For example, acute kidney injury or starting renin–angiotensin system antagonist therapy.
- Be aware that adults with CKD are at increased risk of progression to end-stage renal disease if they have either of the following:
- a sustained decrease in GFR of 25% or more over 12 months or
- a sustained decrease in GFR of 15 ml/min/1.73 m2 or more over 12 months
- When assessing CKD progression, extrapolate the current rate of decline of GFR and take this into account when planning intervention strategies, particularly if it suggests that the person might need renal replacement therapy in their lifetime.
Risk Factors Associated with CKD Progression in Adults
- Work with adults who have any of the following risk factors for CKD progression to optimise their health:
- cardiovascular disease
- proteinuria
- previous episode of acute kidney injury
- hypertension
- diabetes
- smoking
- African, African-Caribbean or Asian family origin
- chronic use of NSAIDs
- untreated urinary outflow tract obstruction.
- In adults with CKD the chronic use of NSAIDs may be associated with progression and acute use is associated with a reversible decrease in GFR. Exercise caution when giving NSAIDs to people with CKD over prolonged periods of time. Monitor the effects on GFR, particularly in people with a low baseline GFR and/or in the presence of other risks for progression.
Information and Education for People with CKD
- Offer people with CKD (and their family members or carers, as appropriate) education and information tailored to the severity and cause of CKD, the associated complications and the risk of progression. For more guidance, see:
- the information on enabling patients to actively participate in their care in NICE’s guideline on patient experience in adult NHS services
- NICE’s guideline on shared decision making
- the section on shared decision making in NICE’s guideline on babies, children and young people’s experience of healthcare
- When developing information or education programmes, involve adults with CKD in their development from the outset. The following topics are suggested.
- What is CKD and how does it affect people?
- What questions should people ask about their kidneys?
- What treatments are available for CKD, what are their advantages and disadvantages, and what complications or side effects may occur as a result of treatment or medication?
- What can people do to manage and influence their own condition?
- In what ways could CKD and its treatment affect people’s daily life, social activities, work opportunities and financial situation, including benefits and allowances available?
- How can people cope with and adjust to CKD and what sources of psychological support are available?
- Information about renal replacement therapy (such as the frequency and length of time of dialysis treatment sessions or exchanges and pre-emptive transplantation) and the preparation needed (such as having a fistula or peritoneal catheter), if appropriate for the person. See NICE’s guideline on renal replacement therapy and conservative management.
- Conservative management and when it may be considered.
- Offer adults with CKD (and their family members or carers, as appropriate) high-quality information or education programmes as appropriate to the severity of their condition to allow time for them to fully understand and make informed choices about their treatment.
- Ensure healthcare professionals providing information and education programmes have specialist knowledge about CKD and the necessary skills to facilitate learning.
- Take account of the psychological aspects of coping with CKD and offer adults with CKD access to support, for example, support groups, counselling or a specialist nurse.
Lifestyle Advice
- Encourage adults with CKD to take exercise, achieve a healthy weight and stop smoking.
Dietary Interventions
- Offer dietary advice about potassium, phosphate, calorie and salt intake appropriate to the severity of CKD.
- If dietary intervention is agreed, provide it alongside education, detailed dietary assessment and supervision to ensure malnutrition is prevented.
Low-protein Diets
- Do not offer low-protein diets (dietary protein intake less than 0.6 to 0.8 g/kg/day) to adults with CKD.
Self-management
- Ensure that systems are in place to:
- inform adults with CKD (and their family members or carers, as appropriate) of their diagnosis
- enable adults with CKD (and their family members or carers, as appropriate) to share in decision making about their care
- support self-management (this includes providing information about blood pressure, smoking cessation, exercise, diet and medicines) and enable adults with CKD to make informed choices.
- Give adults access to their medical data (including diagnosis, comorbidities, test results, treatments and correspondence) through information systems, such as Renal PatientView, to encourage and help them to self-manage their CKD.
Risk Assessment, Referral Criteria and Shared Care
Risk assessment
- Give adults with CKD and their family members or carers (as appropriate) information about their 5-year risk of needing renal replacement therapy (measured using the 4-variable Kidney Failure Risk Equation). Follow NICE’s guideline on shared decision making when communicating risk.
- Use every day, jargon-free language to communicate information on risk. If technical and medical terms are used, explain them clearly.
- Set aside enough time during the consultation to give information on risk assessment and to answer any questions. Arrange another appointment for more discussion if this is needed.
- Document the discussion on risk assessment and any decisions the person makes.
Referral Criteria
- Refer adults with CKD for specialist assessment (taking into account their wishes and comorbidities) if they have any of the following:
- a 5-year risk of needing renal replacement therapy of greater than 5% (measured using the 4-variable Kidney Failure Risk Equation)
- an ACR of 70 mg/mmol or more, unless known to be caused by diabetes and already appropriately treated (see recommendations 1.6.6 and 1.6.7 in the full guideline)
- an ACR of more than 30 mg/mmol (ACR category A3), together with haematuria
- a sustained decrease in eGFR of 25% or more and a change in eGFR category within 12 months
- a sustained decrease in eGFR of 15 ml/min/1.73 m2 or more per year
- hypertension that remains poorly controlled (above the person’s individual target) despite the use of at least 4 antihypertensive medicines at therapeutic doses (see also NICE’s guideline on hypertension in adults)
- known or suspected rare or genetic causes of CKD
- suspected renal artery stenosis.
- Refer children and young people with CKD for specialist assessment if they have any of the following:
- an ACR of 3 mg/mmol or more, confirmed on a repeat early morning urine sample
- haematuria
- any decrease in eGFR
- hypertension
- known or suspected rare or genetic causes of CKD
- suspected renal artery stenosis
- renal outflow obstruction.
- Consider discussing management with a specialist by letter, email, telephone, or virtual meeting, if there are concerns but the person with CKD does not need to see a specialist.
- Refer people with CKD and renal outflow obstruction to urological services, unless urgent treatment is needed (for example, for hyperkalaemia, severe uraemia, acidosis or fluid overload).
Shared Care
- After referral:
- Agree, document and date a care plan with the person with CKD or their family member or carer (as appropriate). Follow:
- the recommendations on patient views and preferences in NICE’s guideline on patient experience in adult NHS services
- NICE’s guideline on shared decision making.
- Agree, document and date a care plan with the person with CKD or their family member or carer (as appropriate). Follow:
- Consider routine follow up at the GP surgery or with a paediatrician rather than in a specialist clinic.
- Specify criteria for future referral and re-referral if GP follow up is agreed. For children and young people, these criteria should be agreed between the GP and secondary care services.
Pharmacotherapy
Blood Pressure Control
See NICE’s guideline on hypertension in adults for advice on blood pressure control in people with frailty and multimorbidity.NICE’s guideline on hypertension in adults recommends using clinic blood pressure for monitoring response to lifestyle changes or medical treatment.
- In adults with CKD and an ACR under 70 mg/mmol, aim for a clinic systolic blood pressure below 140 mmHg (target range 120 to 139 mmHg) and a clinic diastolic blood pressure below 90 mmHg.
- In adults with CKD and an ACR of 70 mg/mmol or more, aim for a clinic systolic blood pressure below 130 mmHg (target range 120 to 129 mmHg) and a clinic diastolic blood pressure below 80 mmHg.
- In children and young people with CKD and an ACR of 70 mg/mol or more, aim for a clinic systolic blood pressure below the 50th percentile for height.
Pharmacotherapy for Hypertension
- Follow the recommendations on treating hypertension in NICE’s guideline on hypertension in adults for adults with CKD, hypertension and an ACR of 30 mg/mmol or less (ACR categories A1 and A2).
- Offer an angiotensin-receptor blocker (ARB) or an angiotensin-converting enzyme (ACE) inhibitor (titrated to the highest licensed dose that the person can tolerate) to adults, children and young people with CKD who have hypertension and an ACR over 30 mg/mmol (ACR category A3 or above).
Pharmacotherapy for CKD in Adults, Children, and Young People with Related Persistent Proteinuria
- For advice on blood glucose control, see:
- NICE’s guideline on type 1 diabetes in adults
- NICE’s guideline on type 2 diabetes in adults
- NICE’s guideline on type 1 and type 2 diabetes in children and young people.
- For adults with CKD and diabetes (type 1 or type 2) offer an ARB or an ACE inhibitor (titrated to the highest licensed dose that the person can tolerate) if ACR is 3 mg/mmol or more.
- For guidance on SGLT2 inhibitors for adults with CKD and type 2 diabetes, see chronic kidney disease in NICE’s guideline on type 2 diabetes in adults.
- For children and young people with CKD and diabetes (type 1 or 2), offer an ARB or an ACE inhibitor (titrated to the highest licensed dose that they can tolerate) if ACR is 3 mg/mmol or more.
- For adults with CKD but without diabetes:
- refer for nephrology assessment and offer an ARB or an ACE inhibitor (titrated to the highest licensed dose that they can tolerate), if ACR is 70 mg/mmol or more
- monitor in line with the recommendations in the section Frequency of monitoring if ACR is above 30 but below 70 mg/mmol; consider discussing with a nephrologist if eGFR declines or ACR increases.
- For children and young people with CKD but without diabetes:
- offer an ARB or an ACE inhibitor if ACR (titrated to the highest licensed dose that they can tolerate) is 70 mg/mol or more
- monitor in line with recommendations in the section Frequency of monitoring if ACR is above 30 but below 70 mg/mmol; consider discussing with a nephrologist if eGFR declines or ACR increases.
- When offering medicines to lower proteinuria to people with frailty, comorbidities or who are taking many other prescribed medicines, follow the recommendations in NICE’s guideline on medicines optimisation to ensure the best possible outcomes. Seek specialist advice if needed, for example from a consultant in care of the elderly, or from a kidney physician if the person asks about contraception.
Renin–Angiotensin System Antagonists
- Do not offer a combination of renin–angiotensin system antagonists to adults with CKD.
- Explain to adults with CKD (and their family members or carers, as appropriate) who are prescribed renin–angiotensin system antagonists about the importance of:
- achieving the optimal tolerated dose of renin–angiotensin system antagonists and
- monitoring eGFR and serum potassium in achieving this safely.
- Measure serum potassium concentrations and estimate the GFR before starting renin–angiotensin system antagonists in people with CKD. Repeat these measurements between 1 and 2 weeks after starting renin–angiotensin system antagonists and after each dose increase.
- Do not routinely offer a renin–angiotensin system antagonist to adults with CKD if their pretreatment serum potassium concentration is greater than 5.0 mmol/litre.
- If an adult cannot use renin–angiotensin system antagonists because of hyperkalaemia:
- assess for and treat any other factors that promote hyperkalaemia and
- recheck serum potassium concentration.
- Be aware that more frequent monitoring of serum potassium concentration may be needed if medicines known to promote hyperkalaemia are prescribed for use in people alongside renin–angiotensin system antagonists.
- Stop renin–angiotensin system antagonists in adults if the serum potassium concentration increases to 6.0 mmol/litre or more and other medicines known to promote hyperkalaemia have been discontinued.
- For recommendations on hyperkalaemia treatment in adults with categories G3b to G5 chronic kidney disease, see NICE’s technology appraisals on sodium zirconium cyclosilicate and patiromer.
- After introducing or increasing the dose of renin–angiotensin system antagonists in adults, do not modify the dose if either:
- the GFR decrease from pretreatment baseline is less than 25% or
- the serum creatinine increase from baseline is less than 30%.
- If there is a decrease in eGFR or increase in serum creatinine after starting or increasing the dose of renin–angiotensin system antagonists, but it is less than 25% (eGFR) or 30% (serum creatinine) of baseline, repeat the test in 1 to 2 weeks. Do not modify the renin–angiotensin system antagonist dose if the change in eGFR is less than 25% or the change in serum creatinine is less than 30%.
- If an adult’s eGFR change is 25% or more, or the change in serum creatinine is 30% or more:
- investigate other causes of a deterioration in kidney function, such as volume depletion or concurrent medication (for example, NSAIDs)
- if no other cause for the deterioration in kidney function is found, stop the renin–angiotensin system antagonist or reduce the dose to a previously tolerated lower dose, and add an alternative antihypertensive medication if needed.
Statins for Adults
- Follow the recommendations in NICE’s guideline on cardiovascular disease: risk assessment and reduction, including lipid modification for the use of statins in adults with CKD.
Oral Antiplatelets and Anticoagulants for Adults
- Offer antiplatelet medicines to adults with CKD for the secondary prevention of cardiovascular disease, but be aware of the increased risk of bleeding.
- For guidance on oral anticoagulants for people with CKD, see NICE’s guidelines on atrial fibrillation and venous thromboembolic diseases.

