Overview
This guideline sets out an antimicrobial prescribing strategy for managing Clostridioides difficile (C. difficile) infection in adults, young people, and children aged 72 hours and over in community settings. It aims to optimise antibiotic use and reduce antibiotic resistance. The recommendations do not cover diagnosis. NICE worked with Public Health England (PHE) to develop this guidance.
The guideline updates existing PHE guidance on treating C. difficile infection, and partially updates NICE’s interventional procedures guidance on faecal microbiota transplant for recurrent C. difficile infection. It updates and replaces Technology Appraisal 601, Medtech Innovation Briefing 247, and Evidence Summaries ES13 and ESNM1.
This Guidelines summary includes a visual summary of the recommendations, and a table to support prescribing decisions.
Visual Summary
Algorithm 1: Clostridioides Difficile Infection: Antimicrobial Prescribing Visual Summary
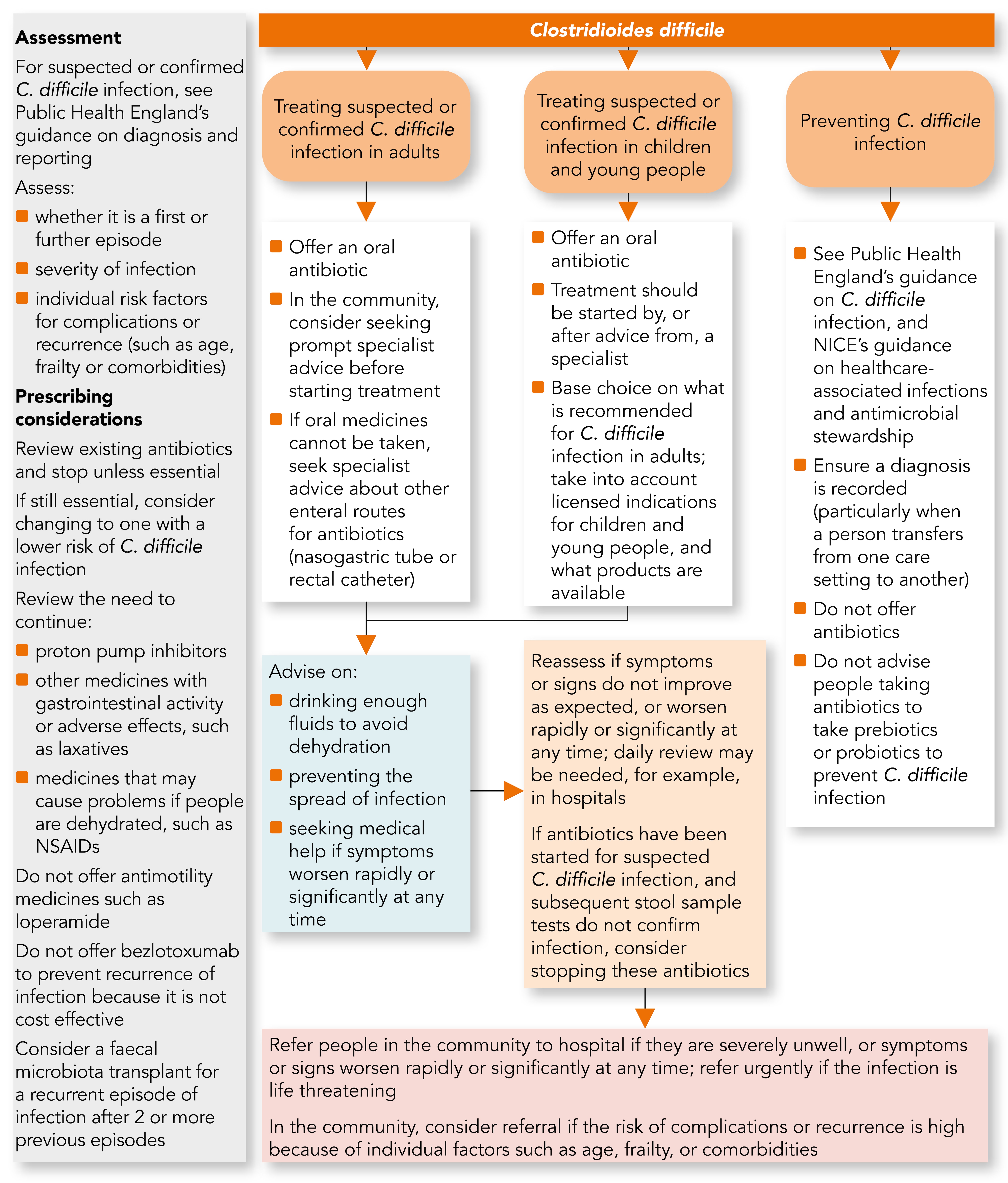
Managing Suspected or Confirmed Clostridioides Difficile Infection
Assessment
- For people with suspected or confirmed C. difficile infection, see PHE’s guidance on diagnosis and reporting.
- For people with suspected or confirmed C. difficile infection, assess:
- whether it is a first or further episode (relapse or recurrence) of C. difficile infection
- the severity of C. difficile infection
- individual factors such as age, frailty or comorbidities that may affect the risk of complications or recurrence.
- For people with suspected or confirmed C. difficile infection, review existing antibiotic treatment and stop it unless essential. If an antibiotic is still essential, consider changing to one with a lower risk of causing C. difficile infection.
- For people with suspected or confirmed C. difficile infection, review the need to continue any treatment with:
- proton pump inhibitors
- other medicines with gastrointestinal activity or adverse effects, such as laxatives
- medicines that may cause problems if people are dehydrated, such as non-steroidal anti-inflammatory drugs, angiotensin-converting enzyme inhibitors, angiotensin-2 receptor antagonists and diuretics.
Treating Suspected or Confirmed C. difficile Infection
- For adults, offer an oral antibiotic to treat suspected or confirmed C. difficile infection (see the recommendations on choice of antibiotic). In the community, consider seeking prompt specialist advice from a microbiologist or infectious diseases specialist before starting treatment.
- For children and young people under 18 years, offer an oral antibiotic to treat suspected or confirmed C. difficile infection. Treatment should be started by, or after advice from, a microbiologist, paediatric infectious diseases specialist or paediatric gastroenterologist.
- For people with suspected or confirmed C. difficile infection who cannot take oral medicines, seek specialist advice from a gastroenterologist or pharmacist about alternative enteral routes for antibiotics, such as a nasogastric tube or rectal catheter.
- Manage fluid loss and symptoms associated with suspected or confirmed C. difficile infection as for acute gastroenteritis. Do not offer antimotility medicines such as loperamide.
- Do not offer bezlotoxumab to prevent recurrence of C. difficile infection because it is not cost effective.
- Consider a faecal microbiota transplant for a recurrent episode of C. difficile infection in adults who have had two or more previous episodes (see NICE’s interventional procedures guidance on faecal microbiota transplant for recurrent C. difficile infection).
Advice
- Advise people with suspected or confirmed C. difficile infection about:
- drinking enough fluids to avoid dehydration
- preventing the spread of infection (see the first recommendation in the section, Preventing C. difficile infection)
- seeking medical help if symptoms worsen rapidly or significantly at any time.
Reassessment
- Reassess people with suspected or confirmed C. difficile infection if symptoms or signs do not improve as expected, or worsen rapidly or significantly at any time. Daily review may be needed, for example, if the person is in hospital.
- If antibiotics have been started for suspected C. difficile infection, and subsequent stool sample tests do not confirm C. difficile infection, consider stopping these antibiotics (see PHE’s guidance on diagnosis and reporting for recommendations on stool sample tests).
Referral
- Refer people in the community with suspected or confirmed C. difficile infection to hospital if they are severely unwell, or their symptoms or signs worsen rapidly or significantly at any time. Refer urgently if the person has a life-threatening infection.
- Consider referring people in the community to hospital if they could be at high risk of complications or recurrence because of individual factors such as age, frailty or comorbidities.
- Ensure that people in hospital with suspected or confirmed C. difficile infection have care from a multidisciplinary team that may include a microbiologist, infectious diseases specialist, gastroenterologist, surgeon, pharmacist or dietitian, as needed.
Choice of Antibiotic
- When prescribing antibiotics for suspected or confirmed C. difficile infection in adults, follow Table 1.
- When prescribing antibiotics for suspected or confirmed C. difficile infection in children and young people under 18 years, base the choice of antibiotic on what is recommended for C. difficile infection in adults. Take into account licensed indications for children and young people, and what products are available (see the BNF for Children for dosing information).
- Use clinical judgement to determine whether antibiotic treatment for C. difficile is ineffective. It is not usually possible to determine this until day 7 because diarrhoea may take 1 to 2 weeks to resolve.
Table 1: Antibiotics for Adults Aged 18 Years and Older
| Treatment | Antibiotic, Dosage, and Course Length |
|---|---|
| First-line antibiotic for a first episode of mild, moderate or severe C. difficile infection | Vancomycin:125 mg orally four times a day for 10 days |
| Second-line antibiotic for a first episode of mild, moderate or severe C. difficile infection if vancomycin is ineffective | Fidaxomicin: 200 mg orally twice a day for 10 days |
| Antibiotics for C. difficile infection if first- and second-line antibiotics are ineffective | Seek specialist advice. Specialists may initially offer: Vancomycin: up to 500 mg orally four times a day for 10 days With or without Metronidazole: 500 mg intravenously three times a day for 10 days |
| Antibiotic for a further episode of C. difficile infection within 12 weeks of symptom resolution (relapse) | Fidaxomicin: 200 mg orally twice a day for 10 days |
| Antibiotics for a further episode of C. difficile infection more than 12 weeks after symptom resolution (recurrence) | Vancomycin: 125 mg orally four times a day for 10 days Or Fidaxomicin: 200 mg orally twice a day for 10 days |
| Antibiotics for life-threatening C. difficile infection (also see the recommendation under Referral in the section, Managing suspected or confirmed Clostridioide difficile infection) | Seek urgent specialist advice, which may include surgery. Antibiotics that specialists may initially offer are: Vancomycin: 500 mg orally four times a day for 10 days With Metronidazole: 500 mg intravenously three times a day for 10 days |
| See the BNF for appropriate use and dosing in specific populations, for example, hepatic impairment, renal impairment, pregnancy and breastfeeding. Also see medicines safety. See Specialist Pharmacy Service guidance on choosing between oral vancomycin options. If ileus is present, specialists may use vancomycin rectally. | |
Preventing C. Difficile Infection
- For how to prevent C. difficile infection through good antimicrobial stewardship, infection control and environmental hygiene measures, see:
- Ensure a diagnosis of C. difficile infection is recorded (particularly when a person transfers from one care setting to another). This is so that it can be taken into account before any future antibiotics are prescribed.
- Do not offer antibiotics to prevent C. difficile infection.
- Do not advise people taking antibiotics to take prebiotics or probiotics to prevent C. difficile infection.

